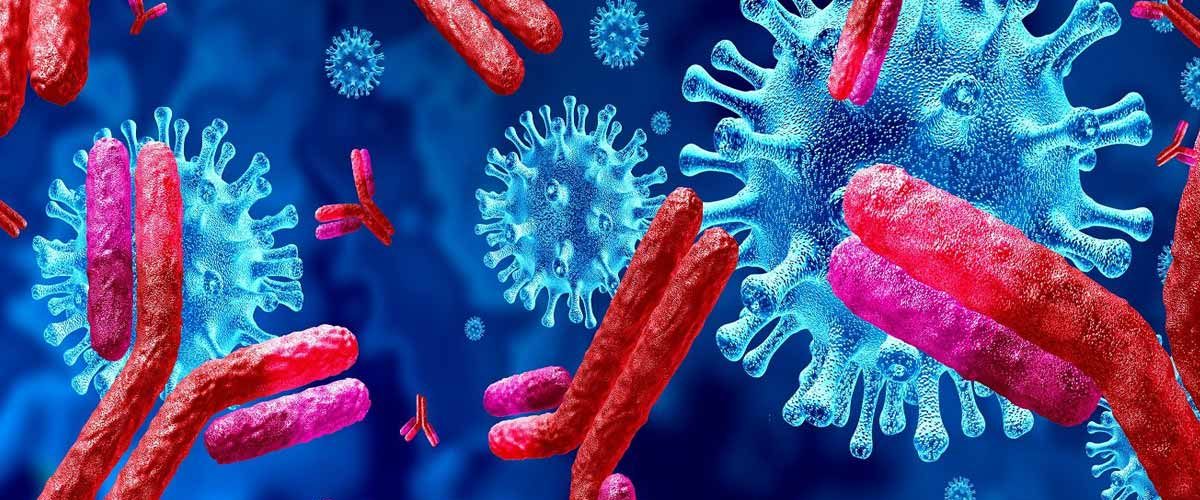
At the German Center for Neurodegenerative Diseases (DZNE) and Charité – Universitätsmedizin Berlin, researchers have successfully identified valuable antibodies to treat the coronavirus SARS-CoV-2 trying to develop a passive vaccination against COVID-19. During this research, they have also revealed that some SARS-CoV-2 antibodies connect to tissue samples from different organs, which may trigger undesired derivative effects. They report their study results in the scholarly journal “Cell.”
Initially, the researchers isolated approximately 600 different antibodies from the blood of individuals who had recovered from COVID-19, the disease caused by SARS-CoV-2. With the help of laboratory testing, they were able to decrease this number to a small digit of antibodies that were predominantly effective at binding to the virus. Subsequently, they artificially produced these antibodies using cell cultures. Crystallographic analysis reveals that the identified presumed neutralizing antibodies bind to the virus and prevent pathogens’ entry and reproduction. Furthermore, antibodies detecting virus helps immune cells to eliminate the pathogen. Research on ‘hamsters’ that are equally susceptible to infection by SARS-CoV-2 Like a human, confirmed the high efficiency of the selected antibodies: “In most of the times, if the antibodies were given after infection, the hamsters developed mild disease signs. On the other hand, when the antibodies were applied preventively even before the occurrence of infection, the animals did not get the infection,” said Dr. Jakob Kreye, director of the current research project. The DZNE researcher is one of the two first authors of the recent publication.
Antibodies for passive vaccination
Antibodies have a long history of treating infectious diseases. For COVID-19, this technique is also being studied by incorporating plasma-derived from the blood of recovered patients. With the plasma, antibodies of donors are transferred. “Superlatively, the most valuable antibody is produced in a controlled method on an industrial scale in stable quality. This is the objective we are pursuing,” said Dr. Momsen Reincke, also the first author of the current publication.
“Three of our antibodies are principally capable of clinical expansion,” explained Prof. Dr. Harald Prüss, a scientist group leader at the DZNE and a senior physician at the Clinic for Neurology with Experimental Neurology at Charité – Universitätsmedizin Berlin. “Employing these antibodies, we have started to formulate a passive vaccination against SARS-CoV-2.” Such a project needs particular assistance from industrial buddies. For that reason, scientists are collaborating with Miltenyi Biotec.
Besides the treatment of patients, precautionary safety measures for healthy individuals who have had direct or indirect contact with infected patients are also potential purpose of the current study. And thus, how long the protection lasts will have to be examined in experimental studies. “In contrasting with active vaccination, the passive vaccination engages the administration of ready-made antibodies, which are degenerated after some time,” Prof. Prüss said a whole passive vaccination provides less persistent protection than that provided by an active vaccination. Although the effect of a passive vaccination is very quick, it has to build up first with an active vaccination. “It is potentially best if both options were accessible so that a flexible response could be generated depending on the condition.”
Modern technologies
Reincke, Kreye, Prüss, and colleagues frequently deal with autoimmune diseases of the brain, in which antibodies inaccurately attack neurons. “In the defiance of the COVID-19 pandemic, though, it was clear to use our resources also in different ways,” said Prof. Prüss. For the current project, the scientists benefit from a project funded by the Helmholtz Association: the “BaoBab Innovation Lab.” Within this agenda, they establish and improve technologies for developing and characterizing antibodies, which they are currently applying. “Now, we are working with our industrial partner to develop the situation that will allow for the most efficient industrial-scale production of the antibodies we have identified,” said Prüss. “The subsequent step is clinical trials that are testing in humans. However, this cannot be predicted before the end of this year at the earliest. The preparation for this has already started.”
Potential side effects
During their research, the scientist made a further discovery: some particularly helpful antibodies against the coronavirus exclusively attached to proteins of the heart muscle, blood vessels, and brain. In trials with tissue samples from mice, several of the neutralizing antibodies showed such cross-reactivity. Therefore, they were not used in the development of a passive vaccination. “These antibodies bind not only to the virus but also to proteins in the body that have nothing to do with the virus. Potential research is needed to investigate whether the related tissues could potentially become targets of attacks by the autoimmune system,” said Prof. Prüss. Whether these clinical results are relevant for humans cannot be predicted at present. “Reasoning from these findings, we need to actively observe and detect any autoimmune reactions that may occur from the perspective of COVID-19 and vaccinations at an early stage. Yet these results can contribute to ensuring the development of an even safer vaccine,” the researchers said.











Add comment