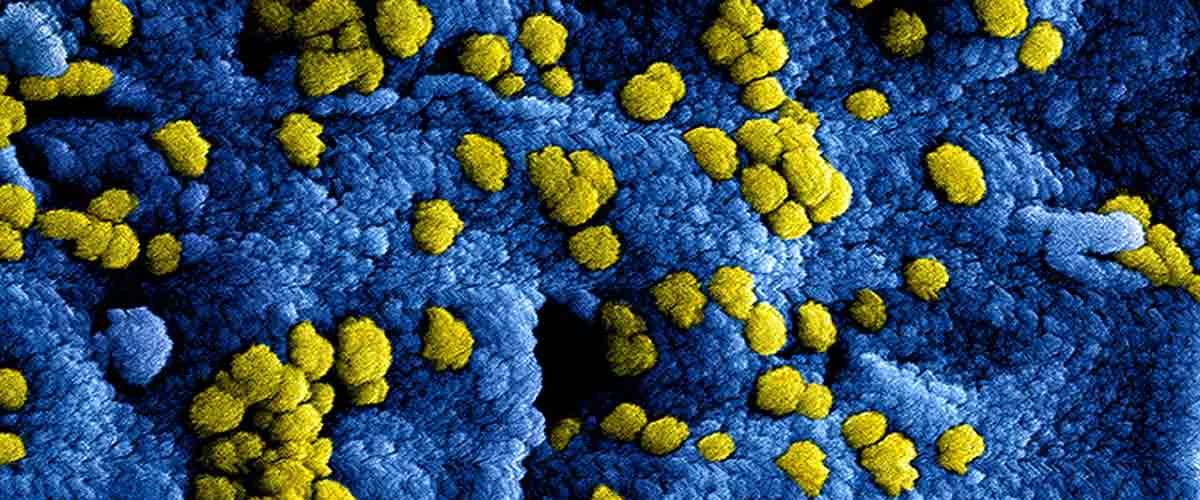
Researchers at “Higher School of Economics University” have discovered microRNA molecules that are potentially able to repress the replication of human coronaviruses, including SARS-CoV-2. It shows that the virus uses miRNA hsa-miR-21-3p to restrain growth at the initial stages of infectivity to detain the active immune response. The findings of the research are published in the journal PeerJ September 14, 2020,
Once the virus enters the cell, it initiates very active interaction with various in-cell molecules. One example of such a molecule class is microRNAs (miRNAs), which are smaller RNAs whose key purpose is to control gene expression. Upon viral entry, miRNAs start binding specific parts of its genome RNA, leading to virus RNAs’ rupture. Such an attack can completely prevent virus replication. Although, in cases when miRNAs are not extremely “aggressive,” such connections do not destroy the virus but rather slow down its replication process. This situation is favorable for the virus since it aids in avoiding a quick immune response in the cell. And some of the viruses persistently accumulate host miRNA binding sites. This turns out to be their advantage: viruses with higher binding sites subsist and replicate better, leading to their developmental domination.
Scientists from the HSE Faculty of Biology and Biotechnology, Stepan Nersisyan, and Alexander Tonevitsky, together with first-year students Narek Engibaryan, Aleksandra Gorbonos, Ksenia Kirdey, and Alexey Makhonin, discovered that cell miRNAs are capable of binding coronavirus genomes.
There is a total of seven types of human coronaviruses. Out of these seven types, four (HCoV-OC43, HCoV-NL63, HCoV-HKU1, and HCoV-229E) are prevalent and cause the illness like common cold, while viruses MERS-CoV, SARS-CoV, and SARS-CoV-2 can cause critical atypical pneumonia. The scientists found four families of human miRNAs with detected binding sites with all the viruses under concern.
To study the virus mode of interaction with the detected miRNAs, the researchers examined the available facts on miRNA sequences in the lungs of mice infected with SARS-CoV. They found out that infectivity leads to an 8-fold increase in the expression of the previously detected miRNA hsa-miR-21-3p.
‘MiRNA hsa-miR-21-3p has great potentiality for binding all human coronaviruses. But following the infection with SARS-CoV, the absorption of this miRNA in the lungs grows a lot. If we believe that this is a form of the immune response, it is uncertain why the virus does not eliminate the binding sites with cell miRNAs in the process of mutation. In contrast, we see that the virus ‘accumulates’ them in its genome during the evolution — our investigation illustrates that such sites are present in all human coronaviruses and do not mutate significantly. We assume that this way, the virus uses this miRNA to slow down its replication in the disease’s initial stages to impediment the active immune response,’ Stepan Nersisyan said. The following step of the researchers will be an experimental examination of their findings. The researchers are also planning to study the prospect of a medicinal effect on the virus that targets the discovered miRNAs. More particular, they plan to find out whether their artificial introduction or elimination can avoid virus reproduction.










Instructive!!
Hopefully, the findings will assist therapists and virologists to develop a possible treatment against COVID-19
Very informative article
These types of studies surely help the scientist to make a permanent cure of Covid-19 disease